1. Introduction
Stability analysis, dispersion relations and pattern formation potential in living tissues, particularly in the perspective of damage and wound healing are analyzed using mathematical frameworks like reaction-diffusion-advection systems and mechanical continuum models. These techniques explain how homogeneous tissues (or uniform wound sites) can break symmetry to form structured, non-uniform patterns like scars, lesions or specific wound healing morphologies. Stability analysis body tissue damage determines whether a system returns to its original equilibrium (stable) or forms a new structure (unstable) after perturbation. Linear stability analysis is adopted where small perturbations are introduced to the equilibrium solutions like normal tissue state and the eigenvalues of the Jacobian matrix are analyzed. If the equilibrium is stable under a reaction-only model (ordinary differential equation system), spatial diffusion and chemotaxis may not necessarily destabilize it
| [14] | Konieczny, L., Roterman-Konieczna, I., Spólnik, P. (2023). The Structure and Function of Living Organisms. In: Systems Biology. Springer, Cham.
https://doi.org/10.1007/978-3-031-31557-2_1 |
| [15] | Maini, P. K. & Woolley, T. E. (2019). The Turing model for biological pattern formation. In: The dynamics of biological systems. SpringeR, 189–204. |
[14, 15]
.
However, in body tissue damage situations, coupling reaction terms (chemical signaling) with diffusion (cell movement) and non-linear damage variables often causes a diffusion-driven unsteadiness. In cases of chronic wound healing, if the basic reproduction number (R
0) of a disease or damage process is greater than 1, then the system will move to a non-trivial (endemic equilibrium) it indicates a sustained, damaged pattern rather than healing
| [17] | Moreo P, Gaffney EA, Garcia-Aznar JM, Doblaré M. (2010). On the modelling of biological patterns with mechanochemical models: insights from analysis and computation. Bull Math Biol. 72(2): 400–431.
https://doi.org/10.1007/s11538-009-9452-4 |
[17]
. The dispersion relation connects the growth rate of perturbations
to their spatial frequency (wave number, 𝑘). It defines the conditions under which patterns emerge. Patterns arise when the dispersion relation
is positive for a range of 𝑘>0, meaning perturbations with certain wavelengths grow for turing stability. In dermal damage models, the spatial arrangement of myofibroblasts and collagen leads to specific damage patterns, such as the dendritic (branching) patterns in dendritic keratitis that is driven by cytokine-induced instabilities for specific tissue patterns. The mechanical models of extracellular matrix (ECM) suggests that fluid-like (viscoelastic) models, such as the Maxwell or Jeffrey models, exhibit a higher pattern formation potential compared to strictly solid-like models
| [9] | Haghighi-Yazdi M. & Lee-Sullivan P (2011) Modeling linear viscoelasticity in glassy polymers using standard rheological models. |
| [11] | Javierre E, Moreo P, Doblaré M.& García-Aznar JM. (2009). Numerical modeling of a mechano–chemical theory for wound contraction analysis. Int J Solids Struct. 46(20): 3597–3606.
https://doi.org/10.1016/j.ijsolstr.2009.06.010 |
[9, 11]
.
The potential for pattern formation in damaged tissues is driven by the interaction of mechanical forces, chemical signals and cellular behaviors. Chronic damage leads to uncontrolled extracellular matrix (ECM) deposition forming scars (fibrosis)
. The "bin or basket weave" pattern of normal tissue breaks down, replaced by aligned collagen fibers that form non-functional structured tissue. The interaction between damaged cells and macrophages can create localized inflammatory spots or waves, analyzed through reaction-diffusion systems. In damage models, the stress-strain state coupled with damage parameters (d∈[0, 1] influences cellular response
| [5] | Chaplain MAJ, V. C.; Gerisch, A. & Lorenzi, T. (2021). Mechanical Models of Pattern and Form in Biological Tissues: The Role of Stress-Strain Constitutive Equations. Bull Math Biol. 83(7): 80. https://doi.org/10.1007/s11538-021-00912-5 |
| [6] | Cross M. & Greenside H. (2009). Pattern formation and dynamics in nonequilibrium systems. Cambridge: Cambridge University Press. |
[5, 6]
. High mechanical stress can either facilitate repair or if beyond thresholds, lead to further tissue damage (necrosis) or apoptosis. Generally, damage occurs to both living and non-living organisms. But this study focuses on tissues in living organisms.
Various researchers added knowledge to this same direction of study were
| [3] | Byrne, H. & Preziosi, L.(2003). Modelling solid tumour growth using the theory of mixtures. Math Med Biol J IMA. 20(4): 341–366. https://doi.org/10.1093/imammb/20.4.341 |
| [9] | Haghighi-Yazdi M. & Lee-Sullivan P (2011) Modeling linear viscoelasticity in glassy polymers using standard rheological models. |
[3, 9]
who carried out their study on mechanical models of pattern and form in biological tissues and the role of stress-strain constitutive equations with findings; that linear stability analysis showed conditions for appearance of pattern and mechanical feedback can influence morphogenesis. The methods used were finite element and ALE methods. Another study on mechanochemical pattern formation in simple models of active viscoelastic fluids and solids was done by the following researchers in the reference number
| [2] | Brinkmann, F., Mercker, M., Richter, T. & Marciniak-Czochra, A. (2018). Post-turing tissue pattern formation: advent of mechanochemistry. PLoS Comput Biol. 14(7): e1006259.
https://doi.org/10.1371/journal.pcbi.1006259 |
| [16] | Maini PK, Woolley TE, Baker RE, Gaffney EA, Lee SS. (2012). Turing’s model for biological pattern formation and the robustness problem. Interface Focus. 2(4): 487–496.
https://doi.org/10.1098/rsfs.2011.0113 |
| [26] | Van Helvert, S. Storm, C. & Friedl, P. (2018). Mechanoreciprocity in cell migration. Nat Cell Biol. 20(1): 8–20.
https://doi.org/10.1038/s41556-017-0012-0 |
[2, 16, 26]
and they came up with the resolution that damaged cells formed voids at on set, later become densely packed as a result of the viscous properties of the cells when evaluated analytically. However, the study of
| [11] | Javierre E, Moreo P, Doblaré M.& García-Aznar JM. (2009). Numerical modeling of a mechano–chemical theory for wound contraction analysis. Int J Solids Struct. 46(20): 3597–3606.
https://doi.org/10.1016/j.ijsolstr.2009.06.010 |
| [17] | Moreo P, Gaffney EA, Garcia-Aznar JM, Doblaré M. (2010). On the modelling of biological patterns with mechanochemical models: insights from analysis and computation. Bull Math Biol. 72(2): 400–431.
https://doi.org/10.1007/s11538-009-9452-4 |
| [20] | Rodrigo, S. G., Hastings, P. J. and Rosenberg, S. M.(2012). Mutation as a Stress Response and the Regulation of Evolvability. HHS Public Access. |
| [24] | Tlili S, Gauquelin E, Li B, Cardoso O, Ladoux B, Delanoë-Ayari H. & Graner F. (2018). Collective cell migration without proliferation: density determines cell velocity and wave velocity. R Soc Open Sci. 5(5): 172421.
https://doi.org/10.1098/rsos.172421 |
[11, 17, 20, 24]
on confinement-induced transition between wavelike collective cell migration modes revealed that the wave vectors was in quadratic form when simulated. In the same vein, the scientists
| [15] | Maini, P. K. & Woolley, T. E. (2019). The Turing model for biological pattern formation. In: The dynamics of biological systems. SpringeR, 189–204. |
| [19] | Petrolli V, Le Goff M, Tadrous M, Martens K, Allier C, Mandula O, Hervé L, Henkes S, Sknepnek R. & Boudou T, et al. (2019). Confinement-induced transition between wavelike collective cell migration modes. Phys Rev Lett. 122(16): 168101. https://doi.org/10.1103/PhysRevLett.122.168101 |
| [22] | Serra-Picamal X, Conte V, Vincent R, Anon E, Tambe DT, Bazellieres E, Butler JP, Fredberg JJ, Trepat X. (2012). Mechanical waves during tissue expansion. Nat Phys. 8(8): 628–634. https://doi.org/10.1038/nphys2355 |
| [23] | Shain, A. H. & Pollack, J. R.(2013). The spectrum of SWI/SNF mutations, ubiquitous in Human Cancer. PLoS ONE. 8(1): e55119. https://doi.org/10.1371/journal.pone.0055119 |
[15, 19, 22, 23]
did a study on structure and function of living organisms using the turing model for biological pattern formation. The results supported that fluid-like constitutive models like Maxwell and Jeffrey models have a pattern formation potential higher than solid-like models of the Kelvin-Voigt model. Therefore, to have more knowledge of this title, the readers would explore these studies
| [1] | Alonso S, Radszuweit M, Engel H. & Bär M. (2017). Mechanochemical pattern formation in simple models of active viscoelastic fluids and solids. J Phys D Appl Phys. 50(43): 434004. https://doi.org/10.1088/1361-6463/aa8a1d |
| [5] | Chaplain MAJ, V. C.; Gerisch, A. & Lorenzi, T. (2021). Mechanical Models of Pattern and Form in Biological Tissues: The Role of Stress-Strain Constitutive Equations. Bull Math Biol. 83(7): 80. https://doi.org/10.1007/s11538-021-00912-5 |
| [6] | Cross M. & Greenside H. (2009). Pattern formation and dynamics in nonequilibrium systems. Cambridge: Cambridge University Press. |
| [7] | Ebihara T, Venkatesan N, Tanaka R, Ludwig MS. (2000). Changes in extracellular matrix and tissue viscoelasticity in bleomycin-induced lung fibrosis: Temporal aspects. Am J Respirat Crit Care Med. 162(4): 1569–1576.
https://doi.org/10.1164/ajrccm.162.4.9912011 |
| [8] | Gilmore SJ, Vaughan BL, Jr, Madzvamuse A. & Maini, PK. (2012). A mechanochemical model of striae distensae. Math Biosci. 240(2): 141–147.
https://doi.org/10.1016/j.mbs.2012.06.007 |
| [12] | Jezierska–Drutel, A., Rosenzweig, S. A. & Neumann, C. A. (2013). Role of Oxidative Stress and the Microenvironment in Breast Cancer Development and Progression. Advances in Cancer Research, Vol 119, Elsevier Inc. |
| [13] | Khalilgharibi N. & Mao Y. (2021). To form and function: on the role of basement membrane mechanics in tissue development, homeostasis and disease. Open Biol. 11(2): 200360.
https://doi.org/10.1098/rsob.200360 |
| [18] | Marray, J. D. (2003). Mathematical Biology II: Spatial Models and Biomedical Applications. 3rd ed. 18(1), Springer New York. |
| [21] | Scianna M, Bell CG, Preziosi L. (2013). A review of mathematical models for the formation of vascular networks. J Theor Biol. 333: 174–209.
https://doi.org/10.1016/j.jtbi.2013.04.037 |
| [25] | Urdy, S. (2012). On the evolution of morphogenetic models: mechano-chemical interactions and an integrated view of cell differentiation, growth, pattern formation and morphogenesis. Biol Rev. 87(4): 786–803.
https://doi.org/10.1111/j.1469-185X.2012.00221.x |
[1, 5-8, 12, 13, 18, 21, 25]
.
2. Materials and Methods
2.1. Assumption
The study considered the general characteristic behaviours of soft tissues in biological systems which Wertheim, (1847) presented force-elongation data for various tissues including arteries and veins that soft tissues do not obey Hooke’s law (i.e. a linear relation between Cauchy stress and a linearized measure of strain). Roy, (1880) detected that arteries exhibit an anisotropic response in an `elastic after-action’ or creep which signifies a viscoelastic character and a thermo elastic behaviour similar to rubber. He added that the material properties of arteries differ with radial location within the wall-a local heterogeneity and along the vascular tree-a regional heterogeneity, that they change with exercise, age, disease and time post-mortem, and even that they differ with gender. This implies that most soft tissues exhibit a nonlinear, inelastic, heterogeneous, anisotropic character that varies from point to point, from time to time and from individual to individual.
From the above structures of soft tissues, the study is looking at stability analysis of damage in the body of living organisms in the area of viscoelastic and thermo elastic constitutive relationship. However, the parameters to be considered are: stress damage relation, strain –damage coupled with constitutive equation and law of elasticity coupled with damage, viscoelastic tensor equation, mechanotaxis equation with mitosis M and conservation equation for the matrix material. From all indications, the model equation will show three equations such as the Force Balance Equation, Cell Conservation Equation and Matrix Conservation Equation respectively.
2.2. Formulation of the Model
Based on the above expositions and assumptions about soft biological tissues, we considered Cell – Matrix Mechanical Interaction Equation which is the mechanical interaction between the cells, matrix and their mechanical damage or deformations. Thus, this model will comprise of material of the cells plus matrix plus cell traction minus body forces as a linear isotropic viscoelastic continuum as functions of the stress tensor. Believing that the traction forces generated by the cells are in mechanical equilibrium with the elastic restoring forces developed in the matrix and the body forces; then the mechanical cell – matrix equation becomes:
(this is called equilibrium force equation)(1)
Where F = the body forces acting on the matrix and is the stress tensor. All the equations in this study has its functional space as position x and time t i.e (x, t). Now the model of stress tensor consists of the ECM and the cell parts; that is
where stress tensor, ExtraCellular Matrix part of the stress and Cell part of the stress.
For soft tissues, a linear viscoelastic material gives the stress–strain constitutive relation below (Landau and Lifshitz 1970);
i.e.(3)
(Viscous part)(4)
Where
,
Hence (
5) is the Extra-Cellular Matrix part of the stress.
Thus, the subscript t denotes partial differentiation with respect to time, I is the unit tensor,
and
are the shear and bulk viscosities of the ECM respectively,
is the strain tensor i.e
; where u is the displacement,
is the dilation and
are the Young’s modulus and Poisson ratio respectively. Modeling in place of the elastic contribution in equation (
3) since fibrous materials are also characterized by nonlocal elastic interaction and fibres can also transmit stress between points in the ECM we have the anisotropic effect and nonlocal effect which can be modeled by writing in place of the elastic contribution in equation
| [17] | Moreo P, Gaffney EA, Garcia-Aznar JM, Doblaré M. (2010). On the modelling of biological patterns with mechanochemical models: insights from analysis and computation. Bull Math Biol. 72(2): 400–431.
https://doi.org/10.1007/s11538-009-9452-4 |
[17]
.
Elastic part)(5)
Where , and and are parameters for long range effects.
If .
Exhibiting the contributions of cell tractions to the stress tensor. This study shows that more cells the greater the traction force; that is to say that cell-cell contact activation while the traction force increases for large damage cell densities. Assuming that the cell traction forces, per unit mass of matrix, initially increases with m and increases with m for very large m; gives:
where is the traction force generated by the cell, n is the number of cells in the matrix and is the measure of force activation resulting from the neighbouring cells. For the cell traction stress () we have:
where
is the cell traction, I is the unit tensor, C is the cell density,
is the number of damage cells transported or pack in an injured area and N (injured steady state) controls the activation of cell traction as the cell density increases. For this study, considering the contribution of
cell to stress tensor
| [12] | Jezierska–Drutel, A., Rosenzweig, S. A. & Neumann, C. A. (2013). Role of Oxidative Stress and the Microenvironment in Breast Cancer Development and Progression. Advances in Cancer Research, Vol 119, Elsevier Inc. |
[12]
yields;
where
is the measure of the nonlocal long range cell –ECM interactions. Considering that the cancer cells are densely packed equation (
9) becomes
is the Stress in the cell
Considering the body force F in equation (
1) which is the activating forces as the body forces are proportional to the density of the ECM and the displacement of the matrix by the numerous injured cells from its strained position to unstrained position gives;
Where
is an elastic parameter characterising the damaged cells or substrate attachments in the body of the organisms giving the model for the force balance equation in (
1) as
(11)
Model equation (
11) shows that there are two other equations that could help in proffering solutions since it has to do with soft tissue and damaged cells in it. Such equations are the cell conservation equation and the matrix conservation equation will be of immense help to this study. Thus, the general form of cell conservation equation is given by
where C is the cell density, J is the flux (instability of the soft tissue damaged cells) of cells, i.e. the number crossing a unit area in a unit time and M is the mitotic or cell proliferation rate. For the damaged cell growth (Mitosis, M) we have
where r is the proliferation rate, is the number of damaged cells in the tissue cell of the organism and N (injured cells) is the maximum cell density. For the convective flux contribution Jc with u (x, t) as the displacement vector of the ECM is given by
Where , is the velocity of deformation of the matrix and, n is the number of damaged cells transported with gross movement of the tissue. We considered that cells disperse in a steady state in an inhomogeneous isotropic medium. Thus, classical diffusion contributes a flux term which models the random (travelling wave motion) motion in which the cells respond to local variations in the cell density and tend to move down the density gradient. This brings in the usual diffusion term as to the conservation equation which represents local or short range motion. Hence, the flux of the cells is given by
Where
is the usual Fickian or short range diffusion coefficient and
is the long range diffusion coefficient and
is the number of damaged cells. The long range diffusion gives rise to a biharmonic term in equation (
15) above.
However, this soft tissue also exhibites a haptotaxis or mechanotaxis movement which is the traction exerted by the cells on the matrix generating gradients in the matrix density
. Considering the density of the matrix with that of adhesive sites for the cell lamellapodia to get hold of the cells free to move in an adhesive gradient tend to move up since the cells can get a stronger grip on the denser matrix. This results to a net flux of cells up the gradient which is assumed to be proportional to
| [7] | Ebihara T, Venkatesan N, Tanaka R, Ludwig MS. (2000). Changes in extracellular matrix and tissue viscoelasticity in bleomycin-induced lung fibrosis: Temporal aspects. Am J Respirat Crit Care Med. 162(4): 1569–1576.
https://doi.org/10.1164/ajrccm.162.4.9912011 |
| [10] | Hylan, KM. (2014.). Tumor suppressor genes and oncogenes: Genes that prevent and cause cancer. |
| [13] | Khalilgharibi N. & Mao Y. (2021). To form and function: on the role of basement membrane mechanics in tissue development, homeostasis and disease. Open Biol. 11(2): 200360.
https://doi.org/10.1098/rsob.200360 |
[7, 10, 13]
. Because of the physical properties of the matrix and nonlocal sensing properties of the cells we bring in a long range effect as in equation (
15) above, then the mechanotaxis flux becomes
Where and are the long range effect coefficients.
Based on the above elucidations and substitution the cell conservation equation is;
(17)
Where are positive parameters, and is the cell density.
Finally for the third equation is the conservation equation for the matrix material which is the matrix density is given by
Where matrix flux is taken to be through convection and is taken to be the rate of secretion or spread in matrix by the damaged cells. Upon expansion with the product rules for vector calculus the model equations becomes:
(20)
The three system of equations for the study model comprises the cell conservation equation which is cell density ; the displacement, and the equation for the matrix material which is the matrix density.
3. Model Analysis
3.1. Equilibrium Analysis
The uniform steady state solutions of (
20) are
and
,
and the equilibrium analysis is evaluated by equating the three nonlinear partial differential equations to zero as
(21)
,,
,and
. So that(22)
To resolve this system of non-linear algebraic equations, we put
,
and
to be the equilibrium solutions for the cell density, displacement and matrix density respectively, then put
, in equation (
22) we have:
(25)
From (
21) considering the fact that matrix has viscous part gives
From the above solutions, one of the equilibria solution is the trivial solution with non-existence of the density fields (cell density and matrix density) and the displacement is.
The other equilibrium solution shows the existence of both cell density and displacement with matrix density satisfied as:
(28)
3.2. Stability Analysis
In solving for the stability, this can be determined by performing a linearization. Considering the complexity in damaged cells reproduction and proliferation in this nonlinear systems of equation by finding the rate of spread,
of damaged cells in the body of the organisms, traction term A, stress term B, diffusion term C, haptotaxis or mechanical term D (movement of the damaged cells) and the secretion term E; all are functions of wave vector in the model equations. Hence, the linear stability of the solution is obtained by seeking solutions of the linearised equations (
20) and writing it in the corresponding variables as below gives.
(29)
(30)
,,,,,,(32)
Since this study is in three dimensional space, we will limit the analysis to direction because we are considering the growth rate and destruction rate of damaged cells, k. Where are in their partial derivatives form and are equal to ,
,,(33)
Thus, considering
,
and
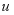
small, upon substituting into the nonlinear system of equation and keeping the linear terms in
,
and
,
, and
is a measure of cell –cell contact activation of cancer cells and also nonnegative. Setting the solutions the linearized equations to be
Where
is the wave vector
and
is the linear growth term. Thus, the dispersion relation
is solutions of the polynomial in
given by the determinant in (
39).
Thus, the Jacobian matrix of the model and their derivatives in the linear system of equations above becomes:
(35)
Upon substitution gives:
(36)
(37)
where is the growth rate (linear growth term) and k is the wavevector. In solving the equations we have to use determinant method which yields:
(39)
In the determinant above, we considered our solutions in the growth rate as a function of rate of destruction of cells , traction factor A, stress term B, diffusion term C, haptotaxis or mechanical term D (movement of the cells) and the secretion term E in the model equations. Thus, putting
,,
so that equation (
39) yields:
(40)
(41)
Similarly,
(42)
For implies that it is not yet an open wound gives the equation below:
and
(46)
Similarly,
and(47)
From equation (40) the traction term A and stress term B yield
(49)
Similarly, expressing (
40) in a quadratic form gives
(50)
Equations (
42), (
43) with
, (
50) and (
51) gives us steady state solutions which means that the secretion surface is activating the rate of growth or damaged cells in the body of the organism. This stable solution is an indication of the presence of damaged cells which is in a steady state growth showing that the dead cells have defiled all medications because of the action of the oxidative stress. These results also established that there is presence of oxidative stress in the injured area which is also acting as an activator to the spread of the injured cells showing that the immune system is becoming weak. This results are in conformity with
| [21] | Scianna M, Bell CG, Preziosi L. (2013). A review of mathematical models for the formation of vascular networks. J Theor Biol. 333: 174–209.
https://doi.org/10.1016/j.jtbi.2013.04.037 |
| [23] | Shain, A. H. & Pollack, J. R.(2013). The spectrum of SWI/SNF mutations, ubiquitous in Human Cancer. PLoS ONE. 8(1): e55119. https://doi.org/10.1371/journal.pone.0055119 |
| [24] | Tlili S, Gauquelin E, Li B, Cardoso O, Ladoux B, Delanoë-Ayari H. & Graner F. (2018). Collective cell migration without proliferation: density determines cell velocity and wave velocity. R Soc Open Sci. 5(5): 172421.
https://doi.org/10.1098/rsos.172421 |
[21, 23, 24]
.
4. Dispersion Relations
4.1. The Spatial Heterogeneous and Homogeneous Solutions of the Linear System
The spatial heterogeneous solutions of the linear system are categorised by a dispersion relation.
. From (
34) and (
41) where
and the algebra gives
as the solutions of
Put and , , and s to replace , , and .
Lemma 1: If
is the dispersion relation and solutions of (
50) with the largest Re
, then we need Re
for
.
Proof:
is the dispersion relation and solutions of (
50) with the largest Re
. Expressing (
39), in a quadratic form in terms of
i.e.
gives
, if(53)
This is the necessary conditions needed for Re for .
Lemma 2: If has Re and this exhibits a range of unstable modes with for .
Proof: From (40) if , then the spatial homogeneous case from
(54)
are the sufficient conditions for all the parameters are positive. Since ; Thence, and the stability is achieved. Thus we two required and satisfied conditions are:
i). to exists at least for some .
ii). to exists at least for some .
The solutions of (
34) with the wave vectors, k are linearly unstable and grow exponentially with respect to time. Similarly, it is expected that these unstable heterogeneous linear solutions will evolve into finite amplitude due to the quadratic curvature which spatially structured solutions. Experientially, it is obvious that the nonlinear system of equations in (
20) with exponentially growth solutions will be bounded due to the
quadratic term in the logistic growth equations and the mitotic rate is set to zero. Therefore the contact inhibition term in the displacement model, u will ensure that the solutions are bounded. This solution is in line with the result obtained from
.
4.2. Complex Dispersion Relations
This section is considering some special cases where one or more factors affecting cell motion or mechanical equilibrium are assumed to be negligible. This can done by setting various parameters to zero and evaluate the result of dispersion relation in (
54). Such parameters as:
(i).
0,
=0,
,
there is no cell diffusion and haptotaxis,
no cell division. The model mechanism (
20) becomes;
(55)
The implication of simple conservation equations for C and m is that the cells and matrix are simply convected by the matrix which is the major transport process and it is seen in network formation. Thus, the one-dimensional form of the model mechanism gives;
(56)
Put , , which linearizes the system to
(57)
From (
20) and (
52), replacing
,
,
and
.
From (
54)
, so the dispersion relation gives
(58)
For Re
,
for some
requires
from (
58)
(59)
In terms of and s becomes;
(60)
This implies that spatial pattern will evolve if and only if
(61)
Thus, the root is not relevant so is for all k. hence, the surface is the bifurcation surface between homogeneity and heterogeneity. In view of the role of cell traction and the form of traction against time and the simulations would be captured in part two of this study.
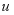 small, upon substituting into the nonlinear system of equation and keeping the linear terms in, and , , and is a measure of cell –cell contact activation of cancer cells and also nonnegative. Setting the solutions the linearized equations to be
small, upon substituting into the nonlinear system of equation and keeping the linear terms in, and , , and is a measure of cell –cell contact activation of cancer cells and also nonnegative. Setting the solutions the linearized equations to be